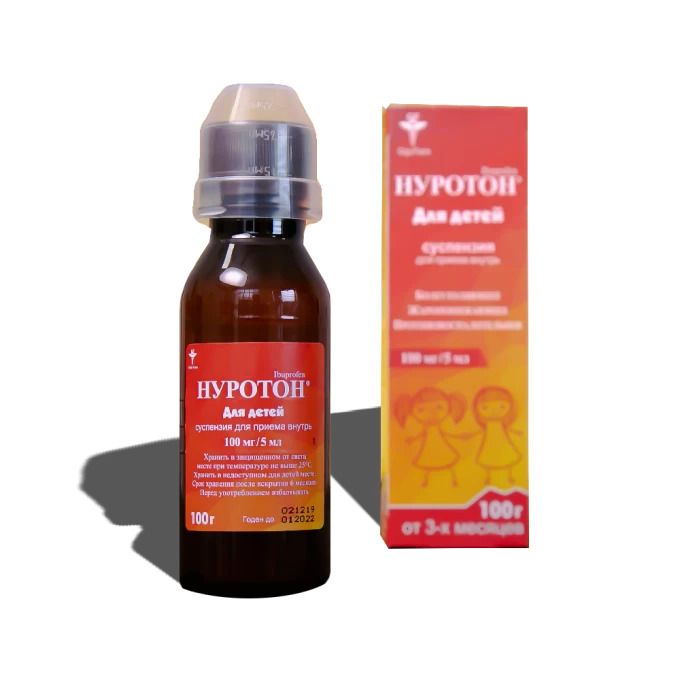
NUROTON SYRUP 100mg 5ml 100g
SHort description
Painkiller, Antipyretic, Anti-inflammatory. For children 100 mg / 5 ml 100 g oral suspension with orange flavor, from 3 months. Composition per 5 ml suspension: ibuprofen 100 mg; liquid maltitol, flavor Orange PH1488, other excipients.
Share
-
Description products
Instruction
on medical use of medicine
NUROTON ®
Dosage form: suspension for intake
Description of dosage form: suspension of white or almost white viscous con-consistency, of sweet taste with a specific smell of orange. Stratification of the suspension is allowed, which is eliminated by shaking.
Composition
5 ml of the suspension contains 100 mg of ibuprofen as an active substance.
auxiliary substances: white crystalline sugar (sucrose), glycerin, propy-lenglikol (E 1520), macrogolglycerol hydroxystearate, citric acid monohydrate, sodium carboxymethylcellulose, anhydrous colloidal silicon dioxide, sodium di-hydrogen phosphate dihydrate, orange flavor, sodium saccharin, methyl parahydroxyben-zoate (E 218), propyl parahydroxybenzoate (E 216), purified water.
Pharmaco-therapeutic group
Non-steroidal anti-inflammatory and antirheumatic drugs. Derivatives of propionic acid.
ATC Code M01AE01
Pharmacological Action
Pharmacodynamic properties
Ibuprofen is a derivative of propionic acid. It has antipyretic, analgesic, anti-inflammatory effect. The mechanism of action of ibuprofen is conditioned, first of all, by the suppression of biosynthesis of prostaglandins (PG) by reducing the activity of cyclooxygenase (COX), an enzyme regulating the conversion of arachidonic acid into prostaglandins, prostacyclin and thromboxane.
In this case, the formation of prostaglandins decreases as a result of irreversible inhibition of the cyclooxygenase pathway of arachidonic acid metabolism. The decrease in the concentration of prostaglandins at the site of inflammation is accompanied by a decrease in the formation of bradykinin, endogenous pyrogens, other biologically active substances, oxygen radicals and NO. All this leads to a decrease in the activity of the inflammatory process (anti-inflammatory effect of ibuprofen) and is accompanied by a decrease in painful reception (analgesic effect). A decrease in the concentration of prostaglandins in the cerebrospinal fluid leads to a normalization of body temperature (antipyretic effect).
Pharmacokinetic properties
After oral administration, more than 80% of ibuprofen is absorbed from the digestive tract.
90% of the drug is absorbed and binds to blood plasma proteins (mainly with albumins). The period of reaching the maximum concentration in blood plasma when taking it on an empty stomach is 45 minutes, when taken after meals - 1,5-2,5 hours; in synovial fluid - 2-3 hours, where higher concentrations are created than in blood plasma. The drug is not accumulated in the body. Ibuprofen is metabolized mainly in the liver. It is subject to presystemic and post-systemic metabolism. After absorption about 60% of the pharmacologically inactive form of ibuprofen is slowly transformed into an active form. 60-90% of the drug is excreted by the kidneys in the form of metabolites and products of their compounds with glucuronic acid. To a lesser extent not more than 1% is released with bile and in an unchanged state. After taking a single dose, the drug is completely excreted within 24 hours.
No special studies of pharmacokinetics have been conducted at children.
The literature data confirm that the absorption, metabolism and excretion of ibuprofen at children occurs in the same way as at adults.Indications for use
· Decrease in body temperature during fever, including post-vaccination.
· Relief of cold and flu symptoms.
· Treatment of pain syndrome from weak to moderate intensity (for example, pain during teething, toothache, earache, headache, pain in case of insignificant injuries and sprains of ligaments and muscles).
Method of Administration and Dosage:
For oral use.
The drug is intended for short-term use only.
5 ml of the suspension contain 100 mg of ibuprofen.
Before use, shake it until a uniform suspension is obtained.
The drug is taken after meals with liquid.
Dosing syringe attached to the package allows you to accurately dose the drug.
1. Open, if available, the individual package of the set, which includes a dosing syringe and an insert, or only a dosing syringe. Remove the cap from the vial. Insert, if necessary, the insert into the neck of the vial so that it is firmly fixed. Insert the dosing syringe into the hole of the insert (the diagram is shown below).
2. Turn the vial with the syringe over and pull the syringe plunger until the plunger tip reaches the mark corresponding to the required volume of the drug (the diagram is shown below). If the required volume of the drug exceeds the maximum volume of the syringe (5 ml), then see point 4.
3. Turn the vial with the syringe to its initial vertical position. Make sure that the syringe plunger has not moved and that the plunger tip is still at the desired mark. Pull the syringe out of the hole in the insert (the insert should remain fixed in the neck of the vial). Close the vial with a cover.
4. The patient should be in a vertical position (except for younger patients, see points 7-10). Slowly push down on the plunger of the syringe, gradually releasing the drug. Sharp pressure on the piston is not allowed. When releasing the drug, it is necessary to direct the opening of the syringe towards the inside of the cheek.
5. If the required volume of the drug exceeds the maximum volume of the syringe (5 ml), then the above points should be repeated so that the total volume of the injected suspension should correspond to the required dose.
5.Disassemble the syringe and rinse it in clean drinking water. Wait for the natural drying of the barrel and piston of the syringe.
6. Keep the vial and dosing syringe out of the reach of children.
The use of the drug at children (up to 3 years), including infants (up to 1 year)
7. After the fulfillment of the abovementioned steps 1-3, place the baby in the feeding position (see figure below).
8. Insert the tip of the syringe into the child's mouth and slowly release the contents towards the inside of the cheek.
9. It is necessary to maintain pauses during the administration of the drug in order to allow the child to gradually swallow the drug.
10. After the child has taken the drug, give him a small amount of water to drink so that the residual amount of the suspension in the oral cavity is swallowed.
11. Follow steps 5-6.
Dosing regime
The likelihood of development of unwilling reactions can be minimized by using the smallest effective dose for the shortest period of time necessary to eliminate the symptoms.
The dose is determined depending on the age and body weight of a child.
It is prescribed at a dose of 20-30 mg / kg / day; this daily dose should be divided into several doses, and a single dose can be from 5 to 10 mg / kg of the child's body weight,It is not recommended for use at children weighing less than 5 kg.
The drug is prescribed in a single dose according to the following scheme:
Age
Body weight
Single dose
Administration order
100mg/5ml
3-6 months
>5 kg
2,5 ml
3 times/day, see also subsection "Other Instructions" under the table
6-12 months
7-9 kg
2,5 ml
up to 3-4 times/day
1-3 years
10-15 kg
5,0 ml
up to 3 times/day
4-6 лет
16-20 kg
7,5 ml
up to 3 times/day
6-9 years
21-30 kg
10,0 ml
up to 3 times/day
9-12 years
31-40 kg
15,0 ml
up to 3 times/day
Children above 12 and adults
More than 40 kg
15,0-20,0 ml
up to 3 times/day
Usage in case of post-vaccinal reactions:
2.5 ml of drug NUROTON ® 100 mg / 5 ml (which corresponds to 50 mg of ibuprofen) once. If necessary, repeated usage of 2.5 ml after 6 hours. It is not allowed to use more than 5 ml of the drug within 24 hours. If the increased body temperature does not decrease, a doctor's consultation is necessary.
Other indications
The interval between the doses should make 6-8 hours (if needed), keeping the period of no less than 4 hours between the next doses.
Do not double the dose if you missed taking the drug! If the next dose is missed, the drug should be taken, if necessary, as soon as you remembered about the omission, under the condition that at least 4 hours have passed since the taking of the previous dose.
The use of the drug to children under 3 months is contraindicated.
The use of the drug by babies from 3- 6 months is possible only after consultation with a doctor. If the symptoms persist for 24 hours (after taking of maximum 3 doses) or worsen at babies aged 3-6 months it is necessary to consult the doctor again.
If the use of the drug at children over 6 months is needed for more than 3 days, or if the symptoms of the disease worsen despite the ongoing treatment, you should consult a doctor.
Do not exceed the recommended dose.
NUROTON is used for no more than 3 days as an antipyretic and no more than 5 days as an anesthetic.
If the fever persists for more than 3 days, and the pain syndrome persists for more than 5 days, you should consult a doctor.
Side effects:
Like other medicines, Nuroton® can cause unwilling effects, although not everybody gets them.
Conditions to pay special attention to (most serious unwilling reactions) are the following:
̶ Immediate allergic reactions
Immediate hypersensitivity reactions may occur when using the drug in rare cases. These reactions occur suddenly, in the very near future (several seconds, minutes) after the use of the drug and can increase rapidly.
The following may appear:
· Anaphylaxis, Quincke's edema or severe shock
Manifestations: one or more of the following symptoms may occur simultaneously - swelling of the larynx, glottis, face, lips, pharynx and / or tongue, which can lead to difficulty of breathing as a result of impaired respiratory tract, difficulty of swallowing or worsening of the ability to speak; often with a simultaneous drop in blood pressure;
· hypersensitivity reactions from the respiratory tract.
Manifestations: Asthma symptoms may occur, worsening of symptoms of pre-existing asthma, bronchospasm, shortness of breath.
If you noticed the occurrence of any of the listed symptoms or other symptoms that, in your opinion, indicate the development of an allergic reaction, you should immediately stop using the drug and immediately turn for medical support.
Severe skin reactions
These reactions can develop during the use or after the end of the use of the drug:
· urticaria.
Manifestations: itchy red rash, accompanied by inflammation of the skin;
· Stevens-Johnson syndrome and toxic epidermal necrolysis (the latter is a more severe form, during which extensive skin peeling occurs, capturing up to 30 percent or more of the body surface).
Manifestations: widespread rash in the form of red spots, blisters, painful ulcers, accompanied by peeling and exfoliation of the skin and mucous membranes; the mucous membranes of the oral cavity, nose, eyes, genitals, any parts of the skin are affected; before the appearance of changes in the skin and mucous membranes, there may be an increase in body temperature, chills, sore throat, headache;
· Acute generalized exenthematous pustulosis.
Manifestations: red scaly widespread rash protruding above the surface of the skin, blisters located mainly on skin folds, trunk and upper limbs, accompanied by fever at the beginning of treatment;
· erythema multiforme;
Manifestations: the appearance of spots in the form of small targets (the dark part in the center is surrounded by a paler zone, a dark ring forms along the contour of the pale zone); other types of spots, vesicles, skin changes similar to blisters may also form; skin lesions may be accompanied by fever and increase of body temperature and increased fatigue, as well as damage of the mucous membranes (eyes, genitals, oral cavity);
· drug reaction with eosinophilia and systemic symptoms (DRESS syndrome).
Manifestations: flu-like symptoms with rash, fever (increase of body temperature), swollen lymph nodes, and changes in blood indices (including the increase of the quantity of eosinophils and the level of liver enzymes).
If you noticed the development of the given symptoms or other symptoms that, in your opinion, indicate the development of a severe skin reaction, you should immediately stop using the drug and consult a doctor immediately, as some of the listed skin reactions can be life-threatening.
Other allergic reactions
Other allergic reactions that may develop during and after the end of the use of the drug are the following:
· other types of skin rash.
You should stop taking the drug immediately and consult a doctor immediately.
The frequency of occurrence of side effects is estimated according to the following scheme: very often (more than 1 person out of 10); often (less than 1 person out of 10); infrequently (less than 1 person out of 100); rarely (less than 1 person out of 1000); very rarely (less than 1 person out of 10,000); frequency is not established (cannot be estimated on the basis of the available data).
Infections and infestations
Very rarely: an exacerbation of the inflammatory process caused by an infection (for example, the development of necrotizing fasciitis), in exceptional cases, severe infectious complications from the skin and soft tissues with chicken pox may occur.
From the gastrointestinal tract:
Infrequently: abdominal pain, nausea, dyspepsia;
Rarely: diarrhea, vomiting, flatulence, constipation;
Very rarely: melena, vomiting with blood, ulcerative stomatitis, gastritis, exacerbation of colitis and Crohn's disease, esophagitis, pancreatitis, the formation of intestinal strictures; the ulceration of the mucous membrane of the stomach and / or duodenum, in some cases complicated by perforation and bleeding, sometimes fatal, especially at the elderly people.
From the liver:
Very rarely: a disorder of the liver function, hepatitis.
From the nervous system:
Infrequently: headache.
Rarely: dizziness, agitation, insomnia, irritability, fatigue. Very rarely: aseptic meningitis (more often at patients with autoimmune diseases, for example at patients with with systemic lupus erythematosus, mixed connective tissue disease), which is manifested by such symptoms as stiff neck muscles, headache, nausea, vomiting, fever, and loss of orientation.
From the organs of vision:
Infrequent: visual disturbances (blurred vision or diplopia), dry and irritated eyes, conjunctival and eyelids edema (allergic genesis).
From the hearing organ
Rarely: hearing loss, ringing in ears or tinnitus
From the blood and lymphatic system
Very rarely: anemia (including hemolytic, aplastic), leukopenia, thrombocytopenia, thrombocytopenic purpura, pancytopenia, agranulocytosis (the first signs of which are fever, sore throat, superficial mouth sores, flu-like symptoms, severe exhaustion, unexplained bleeding and bruising)
From the heart
Very rarely: development or worsening of heart failure, tachycardia. The long-term use of ibuprofen (especially at high doses of 2400 mg per day) may be associated with a slight increase of the risk of arterial thrombosis and pathology caused by arterial thrombosis (myocardial infarction, stroke).
From the vessels
Very rarely: the increase of blood pressure, development of edema. The long-term use of ibuprofen (especially at high doses of 2400 mg per day) may be associated with a slight increase of the risk of arterial thrombosis and pathology caused by arterial thrombosis (myocardial infarction, stroke).
From the kidneys and urinary tract
Very rarely: nephrotic syndrome (edema), acute kidney failure, papillary necrosis, polyuria, increased serum urea concentrations, interstitial nephritis.
From the skin and subcutaneous tissues
Very rarely: alopecia.
If any side effects occur, stop taking the medicine and consult your doctor.
Contraindications
Inform your doctor about any health problems or allergies you have or have had before you start taking this medicine.
- popular individual hypersensitivity to ibuprofen or to other NSAIDs (including to acetylsalicylic acid), as well as auxiliary components of the drug (see section "Composition").)
-Complete or incomplete combination of bronchial asthma, recurrent polyposis of nose and paranasal sinuses and intolerance to acetylsalicylic acid or other NSAIDs (including in anamnesis).
-Erosive and ulcerative diseases of the organs of gastrointestinal tract (including peptic ulcer of the stomach and duodenum in the acute stage, ulcerative colit, peptic ulcer, Crohn's disease).
-Bleeding or perforation in the gastrointestinal tract in anamnesis, associated with the use of NSAIDs.
-Severe kidney failure (creatinine clearance less than 30 ml / min), progressive kidney disease.
- Severe liver failure or active liver disease.
- Severe heart failure.
- Disorders of blood clotting (including hemophilia, prolonged bleeding time, propensity to bleeding, hemorrhagic diathesis);
-The last trimester of pregnancy (see section "Pregnancy and Lactation Period")
- Children's age up to 3 months.
Overdose
You should immediately turn to medical support in case of accidental or intentional ingestion of a large amount of the drug, or if an overdose of the drug is suspected.
At children symptoms may be manifested with oral doses of more than 400 mg / kg. At adults, the dependence of symptoms on the dose is expressed weaker. The half-life of ibuprofen in case of overdose is 1.5-3 hours.
Possible symptoms in case of overdose: nausea, vomiting, abdominal pain, retardation, noise in ears, headache, bleeding from the gastrointestinal tract, drowsiness, violation of the acid-base balance of the blood (development of metabolic acidosis), disorientation in space and / or time (disorientation), coma, hemorrhagic diathesis, bleeding disorder (prolongation of prothrombin time / INR), lowering of blood pressure (hypotension), convulsions, stop of breathing, liver dysfunction, acute kidney failure, decrease of frequency of heart rate (bradycardia), rapid heart rate (tachycardia), "breakdown" of heart rhythm (atrial fibrillation). Children under 5 are especially prone to stop of breathing (apnea), coma and convulsions. Patients with bronchial asthma may have exacerbation of the disease.
Also, in case of an overdose, it is necessary to take into account the presence of the propylene glycol excipient in the composition of the medicine. Propylene glycol on admission
in large quantities has an intoxicating effect, three times less expressed than that of ethanol. Undesirable reactions of propylene glycol during its overdose can be manifested by the central nervous system (oppression of consciousness, coma, convulsions), from the side of the cardiovascular system (decrease in arterial pressure, cardiac arrhythmias). An increase in the osmotic blood pressure (hyperosmolarity) and violation of acid-base balance due to an increase in the level of lactate in the blood (lactacidosis) are most common in patients with kidney failure. Kidney dysfunction (acute tubular necrosis, acute kidney failure), dysfunctions of the respiratory system (respiratory depression, shortness of breath), liver dysfunction, hemolytic reactions (intravascular hemolysis) and the appearance of hemoglobin in the urine (hemoglobinuria) may occur. These adverse reactions are more common with a large ingestion of propylene glycol or with its use at children under 4 years of age, including newborns, pregnant women and patients with liver and kidney failure. Disulfiram-like reactions may also occur in patients receiving simultaneously disulfiram or metronidazole.
Treatment: you should immediately turn for medical support in case of an overdose. It is likely that hospitalization and carrying out of certain activities, such as gastric lavage (only within an hour after taking the drug), taking of activated charcoal, alkaline drinking, symptomatic therapy (correction of the acid-base state, arterial pressure, use of bronchodilators, etc.).
Interaction with other drugs
Inform your doctor if You/ Your child are taking, have recently taken or plan to take any other medicines before You or Your child have been prescribed Nuroton®
Ibuprofen (as well as other non-steroidal anti-inflammatory drugs) should not be used concomitantly with the following medicines.
Acetylsalicylic acid
The simultaneous use of ibuprofen and acetylsalicylic acid is not recommended in connection with the possible increase of the development of undesirable phenomena.
According to the results of laboratory studies, it is supposed that ibuprofen, when used concomitantly with the low doses of acetylsalicylic acid, can competitively inhibit platelet aggregation. Although the admissibility of extrapolation of these data to clinical practice remains uncertain, one cannot exclude the possible effect of regular long-term administration of ibuprofen on reducing the cardioprotective effect of low doses of acetylsalicylic acid. The effect of episodic use of ibuprofen on the cardioprotective properties of acetylsalicylic acid seems unlikely.
Other NSAIDs, including selective inhibitors of cyclooxygenase-2: the simultaneous administration of two or more NSAIDs should be avoided, as the risk of adverse drug effects may increase.
Anticoagulants. NSAIDs may increase the effects of anticoagulants such as warfarin (see section "Special Instructions and Precautions").
Antiplatelet agents and selective inhibitors of serotonin reuptake. When combined with ibuprofen, the risk of gastrointestinal bleeding increases.
Diuretics, ACE inhibitors, beta-adrenoblockers and angiotensin II receptor antagonists. Ibuprofen may reduce the effect of diuretics and other antihypertensive drugs. At some patients with kidney dysfunction, the co-administration of ACE inhibitors, beta-blockers or angiotensin II receptor antagonists and ibuprofen can lead to further worsening of renal function. Thus, the combination of the indicated drugs should be carried out with caution, especially in the elderly.
The simultaneous use of ibuprofen and potassium-sparing diuretics can bring to hyperkalemia.
Digoxin, phenytoin, lithium, methotrexate. The simultaneous use of ibuprofen with digoxin, phenytoin, methotrexate or lithium preparations may increase the concentration of these drugs in the blood. In addition, NSAIDs may worsen the course of heart failure and reduce glomerular filtration rate (GFR) when heart failure is treated with cardiac glycosides.
Cyclosporine. Ibuprofen increases the plasma concentration of cyclosporine and the likelihood of the development of its hepatotoxic effects, as well as increases the risk of development of kidney toxicity.
Tacrolimus. In case of simultaneous administration the risk of nephrotoxicity increases. Corticosteroids. Ibuprofen increases the risk of gastrointestinal bleeding.
Mifepristone. NSAIDs should not be taken within 8-12 days after taking mifepristone because, like all NSAIDs, ibuprofen may bring to reduction of the effect of mifepristone.
Antidiabetic drugs. When used together ibuprofen increases the blood glucose-lowering effect of oral antidiabetic agents (sulfonylurea derivatives) and insulin.
Zidovudine. There is a possibility of an increased risk of hemarthrosis and hematomas in HIV-positive people with hemophilia who are taking zidovudine and ibuprofen simultaneously.
Inductors of microsomal oxidation (phenytoin, ethanol, barbiturates, rifampicin, phenylbutazone, tricyclic antidepressants). They increase the production of hydroxylated active metabolites, increasing the risk of development of severe hepatotoxic reactions.
Antiaids and cholestyramine. They reduce the absorption of ibuprofen. With concomitant prescription the medicines should be administered with an interval of several hours.
Cefamandol, cefoperazone, Iefotetan, valproic acid, plikamycin. They increase the frequency of development of hypoprothrombinemia.
Quinolone antibiotics. The simultaneous use with ibuprofen may increase the risk of development of convulsions.
Captopril. Ibuprofen inhibits sodium excretion (captopril effect).
Probenoid and sulfinpyrazone. Medicines containing probenecid and sulfinpyrazone may delay the release of ibuprofen.
Ritonavir. It can increase the plasma concentrations of ibuprofen.
Aminoglycosides. Ibuprofen can reduce the excretion of aminoglycosides.
Myelotoxic agents. They enhance the manifestation of hematotoxicity of the drug.
Caffeine. It strengthens the analgesic effect of ibuprofen.
SPECIAL INDICATIONS AND PRECAUTIONS
· The severity of unwanted effects can be reduced by taking the minimum effective dose required to control symptoms for the shortest period of time.
· In elderly people, the frequency of the appearance of side reactions caused by NSAIDs is increasing. Gastrointestinal bleeding and perforation are especially dangerous, which can be fatal for the given category of partients. The use of the drug at the given category of patients requires caution and medical supervision.
· The development of bronchospasm is possible at the patients with bronchial asthma or allergic diseases or with such conditions in anamnesis when taking ibuprofen.
· In the presence of systemic lupus erythematosus (SLE) and mixed connective tissue diseases, when taking ibuprofen, the risk of aseptic meningitis is increased.
· Ibuprofen-based preparations should be used with caution by patients with kidney dysfunction, as these disorders may be aggravated by the use of ibuprofen. There is a risk of development of kidney dysfunctions at children with dehydration.
· In case of liver dysfunction NUROTON® is used with caution.
· When treating with ibuprofen in case of the presence of arterial hypertension and / or heart failure in an anamnesis it is necessary to be cautious because of the risk of fluid retention, increase of blood pressure and edema associated with treatment with NSAIDs.
· The results of clinical trials suppose the presence of possible relationship between taking ibuprofen, especially at high doses (≥2400 mg per day), with small increased risk of development of thrombotic phenomena and pathologies associated with thrombosis (for example, myocardial infarction and stroke). Epidemiological studies do not suppose a relationship between the administration of low-dose ibuprofen (≤1200 mg/day) and an increased risk of the development of arterial thrombotic phenomena.
· Influence on blood clotting. Ibuprofen may temporarily inhibit platelets function. Therefore, patients with blood clotting disorders must be carefully examined. The experimental data indicate a decrease in the effect of acetylsalicylic acid as an inhibitor of platelets aggregation when used simultaneously with ibuprofen.
· Patients with uncontrolled arterial hypertension, NYHA class II-III congestive heart failure, established by the ischemic heart disease (IHD), peripheral arterial disease, and/or cerebrovascular diseases, should use ibuprofen only after consulting a doctor, if necessary, under medical supervision, at the same time it is necessary to avoid high doses of ibuprofen (2400 mg/day).
You should consult your doctor on the occasion of possibility of long-term ibuprofen therapy (taking into account the characteristics of your condition), especially by high doses (≥2400 mg per day), in the presence of risk factors for the development of cardiovascular complications (for example, hypertension, hyperlipidemia, diabetes mellitus, smoking).
- The patients having gastrointestinal diseases in the anamnesis (ulcerative colitis, Crohn's disease), should use NSAIDs with caution, as these conditions may worsen.
During the treatment with all NSAIDs, bleeding from the gastrointestinal tract (GIT), the formation of ulcers or their perforation up to death (regardless of the duration of treatment) are possible. In this case, warning symptoms or serious phenomena from the gastrointestinal tract in the anamnesis may be absent. With an increase in the dose of NSAIDs, the risk of bleeding from the gastrointestinal tract, ulceration or perforation increases; also, this risk is higher in patients with ulcer in anamnesis, especially if the ulcer is complicated by bleeding or perforation, and at the elderly people. These patients should start the treatment from the lowest dose.
Patients having toxic effects of NSAIDs on the gastrointestinal tract in the anamnesis should inform the attending physician about all the unusual symptoms in the area of the abdomen (especially about bleeding from the gastrointestinal tract), the elderly people should also pay attention to this in the early stages of treatment.
The patients, concomitantly taking drugs that may increase the risk of ulceration and bleeding, such as oral corticosteroids, anticoagulants (warfarin, etc.), selective serotonin reuptake inhibitors, and antiplatelet agents (aspirin, etc.), should be more cautious.
In case of development of bleeding from the gastrointestinal tract or the formation of peptic ulcer, the patients taking ibuprofen should discontinue the treatment.
Severe skin reactions. On the background of treatment with NSAID serious skin reactions are possible in very rare cases, in some cases with a fatal outcome, including cases of development of exfoliative dermatitis (extensive inflammation and redness of the skin, accompanied by the formation of blisters or peeling of the skin), Stevens-Johnson syndrome and toxic epidermal necrolysis (wide-spread rash in the form of red spots, blisters, painful ulcers, accompanied by peeling and exfoliation of the skin and mucous membranes. The mucous membranes of the mouth, nose, eyes, genitals, any areas of the skin are affected. Before the appearance of changes on the skin and mucous membranes, an increase in body temperature, chills, sore throat, headache may be observed. The latter is a more severe form, in which there is an extensive exfoliation of the skin, capturing up to 30 percent or more of the body surface).
The risk of development of such reactions is more characteristic for the initiation of a course of treatment, in particular, during the first month of therapy. Acute generalized exanthematous pustulosis (red, scaly widespread rash protruding above the surface of the skin, blisters located mainly on the skin folds, trunk and upper limbs, accompanied by fever at the beginning of treatment) has also been reported in case of the use of ibuprofen-containing drugs.
If signs and symptoms of serious skin reactions appear, such as skin rash, mucosal lesions, blisters or any other signs of allergy, the drug should be stopped immediately and a you should consult a doctor.
- There is a possibility that drugs that inhibit cyclooxygenase and prostaglandin synthesis may interfere the female reproductive function by affecting ovulation, which is reversible after treatment is discontinued.
- Excipients. The composition of the drug includes white crystalline sugar (sucrose). Tell your doctor if you have been diagnosed with an intolerance to some sugars before taking this medicine. 5.0 ml of the drug suspension contains 1000.0 mg (0.1 XE) of white crystalline sugar (sucrose), which should be taken into account in patients with diabetes.
The composition of 5.0 ml of the drug includes 85.5 mg of propylene glycol (E 1520).
The medicine contains methyl parahydroxybenzoate (E 218) and propyl parahydroxybenzoate (E 216), which can cause allergic reactions (including the delayed ones).
Masking the infection
NSAIDs may mask the symptoms and signs of infections, such as fever and pain. Therefore, it is possible that Nuroton Zero may delay the appropriate treatment of the infection, which may lead to an increased risk of complication. This has been observed in pneumonia caused by bacteria and bacterial skin infections associated with chickenpox. If Nuroton Zero is used for fever or pain relief due to infection, infection monitoring is recommended. Under non-hospital conditions, the patient should consult a doctor if symptoms persist or worsen.
Pregnancy and lactation
If You are pregnant or breastfeeding, if You do not exclude the possibility of pregnancy at the given moment or if You are planning a pregnancy, please consult Your doctor before starting the use of the drug.
If during the use of Nuroton® You suppose or determine that You are pregnant, consult Your doctor about an alternative treatment. The continued use of Nuroton® during pregnancy may harm Your baby.
Inhibition of prostaglandin synthesis can adversely affect pregnancy. The data from epidemiological studies suppose an increased risk of misbirth, the development of heart disease and a birth defect of the abdominal wall (gastroschisis) in the fetus after the use of prostaglandin synthesis inhibitors in early terms of pregnancy.
It is likely that the risk increases as the dose and duration of drug use increase.
Animal studies with ibuprofen have not shown a teratogenic effect. Nevertheless the use of ibuprofen should be avoided during the first 6 months of pregnancy.
During the third trimester of pregnancy, the use of ibuprofen is contraindicated in connection with the risk of appearance of the following unwilling reactions: the risk of premature closure of the arterial duct of the fetus with possible persistent pulmonary hypertension (up to the development of kidney failure with oligohydramnios), increase in bleeding time at the mother and newborn (antiplatelet effect can be observed even after using very low doses), suppression of uterine contractions (which can lead to a delay of start of birth or make them protracted).
Period of lactation
In small amounts ibuprofen can be excreted with breast milk, therefore it is recommended to stop breastfeeding for the period of treatment.
Effect on ability to drive and other machines
Undesirable effects such as dizziness, drowsiness, fatigue and visual disturbances are possible when taking NSAIDs.
There aren’t any cases of the effect of ibuprofen in the form of a suspension at a dosage of 100 mg / 5 ml on the ability to drive vehicles and mechanisms when using the drug at the recommended doses and for the recommended period of time.
Form of release and package
By 100 gr of suspension in plastic bottles, placed together with a dosing cup and instruction for use in cardboard packs. Each vial, together with a 5.0 ml dosing syringe with an orange or white plunger, an insert for a dosing syringe and instructions for medical use, is placed in a cardboard pack. The insert for the dosing syringe can be inserted into the neck of the vial or enclosed in a pack complete with a dosing syringe. A dosing syringe or a dosing syringe set and an insert for a dosing syringe are placed in a cardboard box in a transparent or white protective individual package, or without package.
STORAGE CONDITIONS:
Store at a temperature not exceeding 25º C, protected from light & moisture
Keep out of the reach of children.
Shelf- life
3 years. The date of expiry is indicated on the package
Do not use it after the expiry date indicated on the package.
Shelf-life after opening is 6 months.
Legal status for supply
Is given without prescription
Manufactured by order of the “Giga Farm” LLC, the Republic of Armenia, t. Gyumri, Gogunts str. 3/5, tel.: +37494000264, +79896335759, e-mail gig.am@mail.ru, www.gigafarm.ru.
Organization receiving claims:
The “Giga Farm” LLC, The Republic of Armenia, t. Gyumri, Gogunts str.3/5.
Tel: +37477 67 61 83. E-mail: Arevik.9187@mail.ru.
Registration certificate holder:
The “Giga Farm” LLC, Republic of Armenia.
“NUROTON®” is a trademark of the “Giga Farm” LLC.
Manufacturer: The “Pharmtechnology” Ltd, the Republic of Belarus, 220024, c. Minsk, Korzhenevskogo street, 22
Instruction
on
medical use of medicine
NUROTON
®
Dosage form: suspension
for intake
Description of dosage form: suspension of white or almost white viscous
con-consistency, of sweet taste with a specific smell of orange. Stratification of the suspension
is allowed, which is eliminated by shaking.
Composition
5 ml of the suspension contains 100 mg
of ibuprofen as an active substance.
auxiliary substances: white crystalline
sugar (sucrose), glycerin, propy-lenglikol (E 1520), macrogolglycerol
hydroxystearate, citric acid monohydrate, sodium carboxymethylcellulose,
anhydrous colloidal silicon dioxide, sodium di-hydrogen phosphate dihydrate,
orange flavor, sodium saccharin, methyl parahydroxyben-zoate (E 218), propyl
parahydroxybenzoate (E 216), purified water.
Pharmaco-therapeutic group
Non-steroidal anti-inflammatory and
antirheumatic drugs. Derivatives of propionic acid.
ATC Code M01AE01
Pharmacological Action
Pharmacodynamic
properties
Ibuprofen is a derivative of propionic acid. It has antipyretic,
analgesic, anti-inflammatory effect. The mechanism of action of ibuprofen is
conditioned, first of all, by the suppression of biosynthesis of prostaglandins
(PG) by reducing the activity of cyclooxygenase (COX), an enzyme regulating the
conversion of arachidonic acid into prostaglandins, prostacyclin and
thromboxane.
In this case, the formation of
prostaglandins decreases as a result of irreversible inhibition of the
cyclooxygenase pathway of arachidonic acid metabolism. The decrease in the
concentration of prostaglandins at the site of inflammation is accompanied by a
decrease in the formation of bradykinin, endogenous pyrogens, other
biologically active substances, oxygen radicals and NO. All this leads to a
decrease in the activity of the inflammatory process (anti-inflammatory effect
of ibuprofen) and is accompanied by a decrease in painful reception (analgesic
effect). A decrease in the concentration of prostaglandins in the cerebrospinal
fluid leads to a normalization of body temperature (antipyretic effect).
Pharmacokinetic
properties
After oral administration, more than
80% of ibuprofen is absorbed from the digestive tract.
90% of the drug is absorbed and binds
to blood plasma proteins (mainly with albumins). The period of reaching the
maximum concentration in blood plasma when taking it on an empty stomach is 45
minutes, when taken after meals - 1,5-2,5 hours; in synovial fluid - 2-3 hours,
where higher concentrations are created than in blood plasma. The drug is not
accumulated in the body. Ibuprofen is metabolized mainly in the liver. It is
subject to presystemic and post-systemic metabolism. After absorption about 60%
of the pharmacologically inactive form of ibuprofen is slowly transformed into
an active form. 60-90% of the drug is excreted by the kidneys in the form of
metabolites and products of their compounds with glucuronic acid. To a lesser
extent not more than 1% is released with bile and in an unchanged state. After
taking a single dose, the drug is completely excreted within 24 hours.
No special studies of pharmacokinetics
have been conducted at children.
The literature data confirm that the absorption, metabolism and excretion of
ibuprofen at children occurs in the same way as at adults.
Indications for use
·
Decrease in
body temperature during fever, including post-vaccination.
·
Relief of cold
and flu symptoms.
·
Treatment of pain
syndrome from weak to moderate intensity (for example, pain during teething, toothache, earache,
headache, pain in case of insignificant injuries and sprains of ligaments and
muscles).
Method of Administration and Dosage:
For oral use.
The
drug is intended for short-term use only.
5 ml of the suspension contain 100 mg
of ibuprofen.
Before use, shake it until a uniform
suspension is obtained.
The drug is taken after meals with
liquid.
Dosing syringe attached to the package
allows you to accurately dose the drug.
1.
Open, if
available, the individual package of the set, which
includes a dosing syringe and an insert, or only a dosing syringe. Remove the cap from the vial. Insert, if necessary, the
insert into the neck of the vial so that it is firmly fixed. Insert the dosing
syringe into the hole of the insert (the diagram is shown below).
2.
Turn the vial
with the syringe over and pull the syringe plunger until the plunger tip
reaches the mark corresponding to the required volume of the drug (the diagram
is shown below). If the required volume of the drug exceeds the maximum volume
of the syringe (5 ml), then see point 4.
3.
Turn the vial
with the syringe to its initial vertical position. Make sure that the
syringe plunger has not moved and that the plunger tip is still at the desired
mark. Pull the syringe out of the hole in the insert (the insert should remain
fixed in the neck of the vial). Close the vial with a cover.
4.
The patient
should be in a vertical position
(except for younger patients, see points 7-10). Slowly push down on the plunger
of the syringe, gradually releasing the drug. Sharp pressure on the piston is
not allowed. When releasing the drug, it is necessary to direct the opening of the syringe towards the inside of the
cheek.
5.
If the required
volume of the drug exceeds the maximum volume of the syringe (5 ml), then the
above points should be repeated so that the total volume of the injected
suspension should correspond to the required dose.
5.Disassemble the syringe and rinse it in clean
drinking water. Wait for the natural drying of the barrel and piston of the
syringe.
6. Keep the vial and dosing syringe out of the reach
of children.
The use of the
drug at children (up
to 3 years), including infants (up to 1 year)
7.
After the fulfillment of the
abovementioned steps 1-3,
place the baby in the feeding position (see figure below).
8.
Insert the tip of the syringe into the child's mouth and
slowly release the contents towards the inside of the cheek.
9.
It is necessary
to maintain pauses during the administration of the drug in order to allow the
child to gradually swallow the drug.
10.
After the child
has taken the drug, give him a small amount of water to drink so that the
residual amount of the suspension in the oral cavity is swallowed.
11.
Follow steps 5-6.
Dosing regime
The likelihood of development of unwilling reactions can be minimized by using the smallest
effective dose for the shortest period of time necessary to eliminate the symptoms.
The dose is determined depending on
the age and body weight of a child.
It is prescribed at a dose of 20-30 mg / kg / day; this daily dose should be
divided into several doses, and a single dose can be from 5 to 10 mg / kg of the child's body weight,
It is not recommended for use at children weighing less than 5 kg.
The drug is prescribed in a single
dose according to the following scheme:
|
Age |
Body weight |
Single dose |
Administration
order |
|
100mg/5ml |
|||
|
3-6 months |
>5 kg |
2,5 ml |
3 times/day, see also subsection
"Other Instructions" under the table |
|
6-12 months |
7-9 kg |
2,5 ml |
up to 3-4 times/day |
|
1-3 years |
10-15 kg |
5,0 ml |
up to 3 times/day |
|
4-6 лет |
16-20 kg |
7,5 ml |
up to 3 times/day |
|
6-9 years |
21-30 kg |
10,0 ml |
up to 3 times/day |
|
9-12 years |
31-40 kg |
15,0 ml |
up to 3 times/day |
|
Children above 12 and adults |
More than 40 kg |
15,0-20,0 ml |
up to 3 times/day |
Usage in case of post-vaccinal reactions:
2.5 ml of drug
NUROTON ® 100 mg / 5 ml (which corresponds to 50 mg of ibuprofen) once. If necessary, repeated usage of 2.5 ml after 6 hours. It is not allowed to use more than 5 ml of the drug within 24 hours. If
the increased body temperature does not decrease, a doctor's
consultation is necessary.
Other indications
The interval between the doses should
make 6-8 hours (if needed), keeping the
period of no less than 4 hours between the next doses.
Do
not double the dose if you missed taking the drug! If the next dose is
missed, the drug should be taken, if necessary, as soon as you remembered about
the omission, under the condition that at least 4 hours have passed since the
taking of the previous dose.
The use of the drug to children under 3 months is contraindicated.
The use of the drug by babies from 3- 6 months is possible only after consultation with a
doctor. If the symptoms persist for 24 hours (after taking of maximum 3 doses) or worsen at babies aged 3-6 months it is necessary
to consult the doctor again.
If the use of the drug at children over 6 months is needed for more than 3 days, or if the symptoms of the
disease worsen despite the ongoing treatment, you should consult a doctor.
Do not exceed the recommended dose.
NUROTON is used for no more than 3 days as an antipyretic and no more than
5 days as an anesthetic.
If the fever persists for more than 3
days, and the pain syndrome persists for more than 5 days, you should consult a
doctor.
Side effects:
Like other medicines, Nuroton®
can cause unwilling effects,
although not everybody gets them.
Conditions
to pay special attention to (most serious unwilling
reactions) are the following:
̶
Immediate allergic reactions
Immediate hypersensitivity reactions may occur when using the drug in rare cases. These reactions occur suddenly, in the
very near future (several seconds, minutes) after the use of the drug and can
increase rapidly.
The following may appear:
·
Anaphylaxis, Quincke's edema or severe shock
Manifestations: one or more of the following symptoms may occur
simultaneously - swelling of the larynx, glottis, face, lips, pharynx and / or
tongue, which can lead to difficulty of breathing as a result of impaired
respiratory tract, difficulty of swallowing or worsening of the ability to speak; often with a simultaneous
drop in blood pressure;
·
hypersensitivity
reactions from the respiratory tract.
Manifestations: Asthma symptoms may occur, worsening of symptoms of pre-existing asthma, bronchospasm,
shortness of breath.
If
you noticed
the occurrence of any of the listed symptoms or other symptoms that, in your
opinion, indicate the development of an allergic reaction, you should
immediately stop using the drug and immediately turn for medical support.
Severe
skin reactions
These reactions can develop during the
use or after the end of the use of the drug:
·
urticaria.
Manifestations: itchy red rash, accompanied by inflammation of the
skin;
·
Stevens-Johnson
syndrome and toxic epidermal necrolysis (the latter is a more severe form, during which extensive skin peeling occurs, capturing up to 30
percent or more of the body surface).
Manifestations: widespread rash in the form of red spots,
blisters, painful ulcers, accompanied by peeling and exfoliation of the skin
and mucous membranes; the mucous membranes of the oral cavity, nose, eyes,
genitals, any parts of the skin are affected; before the appearance of changes
in the skin and mucous membranes, there may be an increase in body temperature,
chills, sore throat, headache;
·
Acute generalized exenthematous pustulosis.
Manifestations: red scaly widespread rash protruding above the
surface of the skin, blisters located mainly on skin folds, trunk and upper
limbs, accompanied by fever at the beginning of treatment;
·
erythema
multiforme;
Manifestations: the appearance of spots in the form of small
targets (the dark part in the center is surrounded by a paler zone, a dark ring
forms along the contour of the pale zone); other types of spots, vesicles, skin
changes similar to blisters may also form; skin lesions may be accompanied by
fever and increase of body temperature and increased
fatigue, as well as damage of the mucous membranes (eyes, genitals, oral cavity);
·
drug reaction
with eosinophilia and systemic symptoms (DRESS syndrome).
Manifestations: flu-like symptoms with rash, fever (increase of body temperature), swollen lymph nodes, and changes in blood indices (including the increase of the quantity of eosinophils and the level of liver enzymes).
If
you noticed the
development of the given symptoms
or other symptoms that, in your opinion, indicate the development of a severe
skin reaction, you should immediately stop using the drug and consult a doctor
immediately, as some of the listed skin reactions can be life-threatening.
Other
allergic reactions
Other allergic reactions that may
develop during and after the end of the use of the drug are the following:
·
other types of
skin rash.
You should stop
taking the drug immediately and consult a doctor immediately.
The frequency
of occurrence of side effects is estimated according to the following scheme:
very often (more than 1 person out of 10); often (less than 1 person out of 10); infrequently (less than 1 person out of 100);
rarely (less than 1 person out of 1000); very
rarely (less than 1 person out of 10,000);
frequency is not established
(cannot be estimated on the basis of the available
data).
Infections and
infestations
Very rarely: an exacerbation of the
inflammatory process caused by an infection (for example, the development of
necrotizing fasciitis), in exceptional cases, severe infectious complications
from the skin and soft tissues with chicken pox may occur.
From the gastrointestinal tract:
Infrequently: abdominal pain, nausea,
dyspepsia;
Rarely: diarrhea, vomiting,
flatulence, constipation;
Very rarely: melena, vomiting with
blood, ulcerative stomatitis, gastritis, exacerbation of
colitis and Crohn's disease, esophagitis, pancreatitis, the formation of
intestinal strictures; the ulceration of the mucous membrane of
the stomach and / or duodenum, in some cases complicated by perforation and
bleeding, sometimes fatal, especially at the elderly people.
From the liver:
Very rarely: a disorder of the liver
function, hepatitis.
From the
nervous system:
Infrequently: headache.
Rarely: dizziness, agitation,
insomnia, irritability, fatigue. Very rarely: aseptic
meningitis (more often at patients with autoimmune diseases, for example at patients with with systemic lupus erythematosus, mixed connective tissue disease), which is manifested by such
symptoms as stiff neck muscles, headache, nausea,
vomiting, fever, and loss of orientation.
From the organs of vision:
Infrequent: visual disturbances
(blurred vision or diplopia), dry and irritated eyes, conjunctival and eyelids
edema (allergic genesis).
From the hearing organ
Rarely: hearing loss, ringing in ears
or tinnitus
From the
blood and lymphatic
system
Very rarely: anemia (including hemolytic, aplastic), leukopenia, thrombocytopenia,
thrombocytopenic purpura, pancytopenia, agranulocytosis (the first signs of which are fever, sore throat,
superficial mouth sores, flu-like symptoms, severe exhaustion, unexplained
bleeding and bruising)
From the heart
Very rarely: development or worsening of
heart failure, tachycardia. The long-term use of ibuprofen (especially at high
doses of 2400 mg per day) may be associated with a slight increase of the risk
of arterial thrombosis and pathology caused by arterial thrombosis (myocardial
infarction, stroke).
From the vessels
Very rarely: the increase of blood pressure, development of edema. The
long-term use of ibuprofen (especially at high doses of 2400 mg per day) may be
associated with a slight increase of the risk of arterial thrombosis and
pathology caused by arterial thrombosis (myocardial infarction, stroke).
From the kidneys and urinary tract
Very rarely: nephrotic syndrome (edema), acute kidney failure, papillary
necrosis, polyuria, increased serum urea concentrations, interstitial
nephritis.
From the skin and subcutaneous tissues
Very rarely: alopecia.
If any side effects occur, stop taking the medicine and
consult your doctor.
Contraindications
Inform your
doctor about any health problems or allergies you have or have had before you
start taking this medicine.
- popular individual hypersensitivity to ibuprofen or to other NSAIDs (including to acetylsalicylic acid), as well as auxiliary components of
the drug (see section "Composition").)
-Complete or incomplete combination of bronchial asthma, recurrent
polyposis of nose and paranasal sinuses and intolerance to acetylsalicylic acid
or other NSAIDs (including in anamnesis).
-Erosive and ulcerative diseases of the organs of gastrointestinal tract
(including peptic ulcer of the stomach and duodenum in the acute stage,
ulcerative colit, peptic ulcer, Crohn's disease).
-Bleeding or perforation in the gastrointestinal tract in anamnesis,
associated with the use of NSAIDs.
-Severe kidney failure (creatinine clearance less than 30 ml / min),
progressive kidney disease.
- Severe liver failure or active liver disease.
- Severe heart failure.
- Disorders of blood clotting
(including hemophilia, prolonged bleeding time, propensity to bleeding,
hemorrhagic diathesis);
-The last trimester of pregnancy (see section
"Pregnancy and Lactation Period")
- Children's age up to 3 months.
Overdose
You should immediately turn to medical support in case of accidental or intentional
ingestion of a large amount of the drug, or if an overdose of the drug is
suspected.
At children symptoms may be manifested
with oral doses of more than 400 mg / kg. At adults, the dependence of symptoms
on the dose is expressed weaker. The half-life of ibuprofen in case of overdose
is 1.5-3 hours.
Possible symptoms in case of overdose: nausea,
vomiting, abdominal pain, retardation, noise in ears, headache, bleeding from
the gastrointestinal tract, drowsiness, violation of
the acid-base balance of the blood (development of metabolic acidosis), disorientation in space and / or time
(disorientation), coma, hemorrhagic diathesis, bleeding disorder (prolongation of prothrombin time / INR), lowering of blood pressure (hypotension), convulsions, stop of breathing, liver dysfunction, acute
kidney failure, decrease of frequency of heart rate (bradycardia), rapid heart
rate (tachycardia), "breakdown" of heart rhythm
(atrial fibrillation). Children under 5 are especially prone to stop of breathing (apnea), coma and convulsions. Patients
with bronchial asthma may have exacerbation of the disease.
Also, in case of an overdose, it is
necessary to take into account the presence of the propylene glycol excipient in the
composition of the medicine. Propylene glycol on
admission
in large quantities has an intoxicating effect,
three times less expressed than that of
ethanol. Undesirable reactions of propylene glycol during its overdose can be manifested by the central nervous system (oppression of consciousness, coma, convulsions), from the side of the
cardiovascular system (decrease in arterial pressure, cardiac arrhythmias). An increase in the
osmotic blood pressure (hyperosmolarity) and
violation of acid-base balance due to an increase in the level of lactate in
the blood (lactacidosis) are most common in patients with kidney failure. Kidney dysfunction (acute tubular necrosis, acute kidney failure), dysfunctions of the respiratory system (respiratory
depression, shortness of breath), liver dysfunction, hemolytic reactions
(intravascular hemolysis) and the appearance of hemoglobin in the urine
(hemoglobinuria) may occur. These adverse reactions are more common with a large ingestion of
propylene glycol or with its use at children under 4 years of age,
including newborns, pregnant women and patients with liver and kidney failure. Disulfiram-like reactions may
also occur in patients receiving simultaneously disulfiram or metronidazole.
Treatment: you should immediately turn for
medical support in case of an
overdose. It is likely that hospitalization and carrying out of certain activities,
such as gastric lavage (only within an hour after taking the drug), taking of
activated charcoal, alkaline drinking,
symptomatic therapy (correction of the acid-base state, arterial
pressure, use of bronchodilators, etc.).
Interaction with other drugs
Inform your
doctor if You/ Your child are taking, have recently taken or plan
to take any other medicines before You or Your child have been prescribed Nuroton®
Ibuprofen (as well as other
non-steroidal anti-inflammatory drugs) should not be used concomitantly with the following medicines.
Acetylsalicylic acid
The simultaneous use of ibuprofen and
acetylsalicylic acid is not recommended in connection with the possible
increase of the development of undesirable phenomena.
According to the results of laboratory
studies, it is supposed that ibuprofen, when used concomitantly with the low
doses of acetylsalicylic acid, can competitively inhibit platelet aggregation.
Although the admissibility of extrapolation of these data to clinical practice
remains uncertain, one cannot exclude the possible effect of regular long-term
administration of ibuprofen on reducing the cardioprotective effect of low
doses of acetylsalicylic acid. The effect of episodic use of ibuprofen on the
cardioprotective properties of acetylsalicylic acid seems unlikely.
Other NSAIDs, including
selective inhibitors of cyclooxygenase-2: the simultaneous administration of two or more NSAIDs should be
avoided, as the risk of adverse drug effects may increase.
Anticoagulants. NSAIDs may increase the effects of
anticoagulants such as warfarin (see section "Special Instructions and Precautions").
Antiplatelet
agents and selective inhibitors of serotonin
reuptake. When combined
with ibuprofen, the risk of gastrointestinal bleeding increases.
Diuretics, ACE inhibitors,
beta-adrenoblockers and angiotensin II receptor antagonists. Ibuprofen may reduce the effect of diuretics and
other antihypertensive drugs. At some patients with kidney dysfunction, the co-administration
of ACE inhibitors, beta-blockers or angiotensin II receptor antagonists and
ibuprofen can lead to further worsening of renal function. Thus, the
combination of the indicated drugs should be carried out with caution,
especially in the elderly.
The simultaneous use of ibuprofen and
potassium-sparing diuretics can bring to hyperkalemia.
Digoxin, phenytoin, lithium, methotrexate. The simultaneous use of ibuprofen with digoxin,
phenytoin, methotrexate or lithium preparations may increase the concentration
of these drugs in the blood. In addition, NSAIDs may worsen the course of heart failure and reduce glomerular filtration rate
(GFR) when heart failure is treated with cardiac glycosides.
Cyclosporine. Ibuprofen increases the plasma concentration of cyclosporine and the
likelihood of the development of its hepatotoxic effects, as well as increases
the risk of development of kidney toxicity.
Tacrolimus. In case of simultaneous administration the risk of nephrotoxicity
increases. Corticosteroids.
Ibuprofen increases the risk of gastrointestinal bleeding.
Mifepristone. NSAIDs should not be taken within 8-12 days after taking mifepristone because, like all NSAIDs,
ibuprofen may bring to reduction of the effect of mifepristone.
Antidiabetic drugs. When used together ibuprofen
increases the blood glucose-lowering effect of oral antidiabetic agents
(sulfonylurea derivatives) and insulin.
Zidovudine. There is a possibility of an increased risk of hemarthrosis and
hematomas in HIV-positive people with hemophilia who are taking zidovudine and
ibuprofen simultaneously.
Inductors of microsomal oxidation (phenytoin,
ethanol, barbiturates, rifampicin, phenylbutazone, tricyclic antidepressants). They increase the production of hydroxylated
active metabolites, increasing the risk of development of severe hepatotoxic
reactions.
Antiaids and cholestyramine. They reduce the absorption of ibuprofen. With
concomitant prescription the medicines
should be administered with an interval of several hours.
Cefamandol,
cefoperazone, Iefotetan, valproic acid, plikamycin. They increase the
frequency of development of hypoprothrombinemia.
Quinolone antibiotics. The simultaneous use with ibuprofen may increase
the risk of development of convulsions.
Captopril. Ibuprofen inhibits sodium excretion (captopril effect).
Probenoid and sulfinpyrazone. Medicines containing probenecid and sulfinpyrazone
may delay the release of ibuprofen.
Ritonavir. It can
increase the plasma concentrations of ibuprofen.
Aminoglycosides. Ibuprofen can reduce the excretion of
aminoglycosides.
Myelotoxic agents. They enhance the manifestation of hematotoxicity
of the drug.
Caffeine. It
strengthens the analgesic effect of ibuprofen.
SPECIAL
INDICATIONS AND
PRECAUTIONS
·
The severity of
unwanted effects can be reduced by taking the minimum effective dose required
to control symptoms for the shortest period of time.
·
In elderly
people, the frequency of the appearance of side reactions caused
by NSAIDs is increasing. Gastrointestinal bleeding and perforation are especially dangerous, which can be
fatal for the given
category of partients. The use of the
drug at the given category of patients requires caution and medical
supervision.
·
The development of bronchospasm is possible at the patients with bronchial asthma or allergic
diseases or with such
conditions in anamnesis when taking ibuprofen.
·
In the presence
of systemic lupus
erythematosus (SLE) and mixed connective tissue diseases, when taking ibuprofen, the risk of aseptic
meningitis is increased.
·
Ibuprofen-based
preparations should be used with caution by patients
with kidney dysfunction, as these disorders may be aggravated by the use of
ibuprofen. There is a risk of development of kidney dysfunctions at children with dehydration.
·
In case of liver
dysfunction NUROTON® is
used with caution.
·
When treating with
ibuprofen in case of the presence of arterial hypertension and / or heart failure in an anamnesis it is necessary to be cautious because of the risk of fluid retention,
increase of blood pressure and edema associated
with treatment with NSAIDs.
·
The results of
clinical trials suppose the presence of possible relationship between taking ibuprofen,
especially at high doses (≥2400 mg per day), with small increased risk of development of thrombotic phenomena and pathologies associated with
thrombosis (for example,
myocardial infarction and stroke). Epidemiological studies do not suppose a relationship between the administration of low-dose ibuprofen (≤1200 mg/day) and an increased
risk of the development of arterial
thrombotic phenomena.
·
Influence on
blood clotting. Ibuprofen may temporarily inhibit platelets function. Therefore, patients
with blood clotting disorders must be
carefully examined. The experimental data indicate a decrease
in the effect of acetylsalicylic acid as an inhibitor of platelets aggregation when used simultaneously with ibuprofen.
·
Patients with uncontrolled arterial hypertension, NYHA class II-III
congestive heart failure, established by the ischemic
heart disease (IHD),
peripheral arterial disease, and/or cerebrovascular
diseases, should use ibuprofen only after
consulting a doctor, if necessary, under medical supervision, at the same time it is
necessary to avoid high doses of ibuprofen (2400
mg/day).
You should consult your doctor on the occasion of possibility of long-term ibuprofen therapy (taking into
account the characteristics of your condition), especially by high doses (≥2400 mg per day), in the presence of risk
factors for the development of cardiovascular complications (for example,
hypertension, hyperlipidemia, diabetes mellitus, smoking).
-
The patients having gastrointestinal diseases in the anamnesis (ulcerative colitis, Crohn's
disease), should use NSAIDs with
caution, as these conditions may worsen.
During the treatment with all NSAIDs, bleeding from the gastrointestinal tract
(GIT), the formation of ulcers or their perforation up to death (regardless of
the duration of treatment) are possible. In this case, warning symptoms or
serious phenomena from the
gastrointestinal tract in the anamnesis may be absent. With an increase in the
dose of NSAIDs, the risk of bleeding from the gastrointestinal tract,
ulceration or perforation increases; also, this risk is higher in patients with
ulcer in anamnesis, especially if the ulcer is complicated by bleeding
or perforation, and at the elderly people. These
patients should start the treatment from the lowest dose.
Patients
having toxic
effects of NSAIDs on the gastrointestinal tract in the anamnesis should inform the attending physician
about all the unusual
symptoms in the area
of the abdomen (especially about bleeding from the gastrointestinal tract), the
elderly people should also pay
attention to this in the early stages of treatment.
The patients, concomitantly taking drugs
that may increase the risk of ulceration and bleeding, such as oral
corticosteroids, anticoagulants (warfarin, etc.), selective serotonin reuptake
inhibitors, and antiplatelet agents (aspirin, etc.), should be more cautious.
In
case of development of bleeding from the gastrointestinal tract or the
formation of peptic ulcer, the patients taking ibuprofen should discontinue the
treatment.
Severe skin
reactions. On the background of treatment with NSAID serious
skin reactions are possible in very rare cases, in some cases with a fatal
outcome, including cases of development of exfoliative dermatitis (extensive
inflammation and redness of the skin, accompanied by the formation of blisters
or peeling of the skin), Stevens-Johnson syndrome and toxic epidermal
necrolysis (wide-spread rash in the form of red spots, blisters, painful
ulcers, accompanied by peeling and exfoliation of the skin and mucous
membranes. The mucous membranes of the mouth, nose, eyes, genitals, any areas
of the skin are affected. Before the appearance of changes on the skin and
mucous membranes, an increase in body temperature, chills, sore throat,
headache may be observed. The latter is a more severe form, in which there is
an extensive exfoliation of the skin, capturing up to 30 percent or more of the
body surface).
The risk of development of such
reactions is more characteristic for the initiation of a course of treatment,
in particular, during the first month of therapy. Acute
generalized exanthematous pustulosis (red, scaly widespread rash protruding
above the surface of the skin, blisters located mainly on the skin folds, trunk
and upper limbs, accompanied by fever at the beginning of treatment) has also
been reported in case of the use of ibuprofen-containing drugs.
If
signs and symptoms of serious skin reactions appear, such as skin rash, mucosal
lesions, blisters or any other signs of allergy, the drug should be stopped
immediately and a you should consult a doctor.
-
There is a
possibility that drugs that inhibit cyclooxygenase and prostaglandin synthesis
may interfere the female reproductive function by
affecting ovulation, which is reversible after treatment is discontinued.
-
Excipients.
The composition of the drug includes white crystalline
sugar (sucrose). Tell your doctor if you have been diagnosed with an
intolerance to some sugars before taking this medicine. 5.0 ml of the drug
suspension contains 1000.0 mg (0.1 XE) of white crystalline sugar (sucrose), which should
be taken into account in patients with diabetes.
The composition of 5.0 ml of the drug
includes 85.5 mg of propylene glycol (E 1520).
The medicine contains methyl
parahydroxybenzoate (E 218) and propyl parahydroxybenzoate (E 216), which can
cause allergic reactions (including the delayed ones).
Masking
the infection
NSAIDs may mask the symptoms and signs
of infections, such as fever and pain. Therefore, it is possible that Nuroton Zero
may delay the appropriate treatment of the infection, which may lead to an
increased risk of complication. This has been observed in pneumonia caused by
bacteria and bacterial skin infections associated with chickenpox. If Nuroton
Zero is used for fever or pain relief due to infection, infection monitoring is
recommended. Under non-hospital conditions, the patient should consult a doctor
if symptoms persist or worsen.
Pregnancy
and lactation
If You are pregnant or breastfeeding, if
You do not exclude the possibility of pregnancy at the given moment or if You
are planning a pregnancy, please consult Your doctor before starting the use of
the drug.
If during the use of Nuroton® You suppose
or determine that You are pregnant, consult Your doctor about an alternative
treatment. The continued use of Nuroton® during pregnancy may harm Your baby.
Inhibition of prostaglandin synthesis can adversely affect
pregnancy. The data
from epidemiological studies suppose
an increased risk of misbirth,
the development of heart disease and a birth defect of the abdominal wall
(gastroschisis) in the fetus after
the use of prostaglandin synthesis inhibitors in early terms of pregnancy.
It is likely that the risk increases as the dose and duration of
drug use increase.
Animal
studies with ibuprofen have not shown a teratogenic effect. Nevertheless the use of ibuprofen should be avoided
during the first 6 months of pregnancy.
During the third trimester of
pregnancy, the use of ibuprofen is contraindicated in connection
with the risk of appearance of the following unwilling reactions: the risk of premature closure of the arterial duct
of the fetus with possible persistent pulmonary hypertension (up to the development of
kidney failure with oligohydramnios), increase in bleeding time at the mother
and newborn (antiplatelet effect can be observed even after using very low
doses), suppression of uterine contractions (which can lead to a delay of start
of birth or make them protracted).
Period of lactation
In small amounts ibuprofen can be
excreted with breast milk, therefore it is recommended to stop breastfeeding
for the period of treatment.
Effect on ability to drive and other machines
Undesirable
effects such as dizziness, drowsiness, fatigue and visual disturbances are
possible when taking NSAIDs.
There aren’t any cases of the effect
of ibuprofen in the form of a suspension at a dosage of 100 mg / 5 ml on the
ability to drive vehicles and mechanisms when using the drug at the recommended
doses and for the recommended period of time.
Form of release and package
By 100 gr of suspension in plastic
bottles, placed together with a dosing cup and instruction for use in cardboard
packs. Each vial, together with a 5.0 ml dosing syringe with an orange or white
plunger, an insert for a dosing syringe and instructions for medical use, is
placed in a cardboard pack. The insert for the dosing syringe can be inserted
into the neck of the vial or enclosed in a pack complete with a dosing syringe.
A dosing syringe or a dosing syringe set and an insert for a dosing syringe are
placed in a cardboard box in a transparent or white protective individual
package, or without package.
STORAGE
CONDITIONS:
Store at
a temperature not exceeding 25º C, protected from light & moisture
Keep out
of the reach of children.
Shelf- life
3 years. The date of expiry is indicated on the
package
Do not use it after the expiry date indicated on the package.
Shelf-life after opening is 6 months.
Legal status for supply
Is given without prescription
Manufactured by order of the “Giga Farm” LLC,
the Republic of Armenia, t. Gyumri, Gogunts str. 3/5, tel.: +37494000264,
+79896335759, e-mail gig.am@mail.ru, www.gigafarm.ru.
Organization
receiving claims:
The “Giga Farm” LLC, The Republic of
Armenia, t. Gyumri, Gogunts str.3/5.
Tel: +37477 67 61 83. E-mail: Arevik.9187@mail.ru.
Registration
certificate holder:
The “Giga Farm” LLC, Republic of Armenia.
“NUROTON®” is a trademark of the “Giga Farm” LLC.
Manufacturer: The “Pharmtechnology”
Ltd, the Republic of Belarus, 220024, c. Minsk, Korzhenevskogo street, 22